Any content you receive is for information purposes only. Always conduct your own research.*Sponsored
Krypton Street Just Put Jaguar Health, Inc. (Nasdaq: JAGX) On Its Watchlist This Morning—Wednesday, April 8, 2026
Don’t Miss The Next Breakout—Get Real-Time Alerts Sent Directly To Your Phone. Up To 10X Faster Than Email.
Our Coverage On (JAGX) is Kicking Off Right Now
Put (JAGX) On Your Screen While It’s Still Early…
April 8, 2026
Momentum Watch | (Nasdaq: JAGX) Makes an Approx. 18% Move on Headline
Dear Reader, Breaking news crossed the wire this morning—and it’s quickly drawing attention to a name that had already been gaining momentum behind the scenes. When a company reaches a key milestone tied to regulatory progression and upcoming data, it can quickly shift how the story is viewed. That’s exactly what we’re seeing unfold right now. Jaguar Health, Inc. (Nasdaq: JAGX) just announced it has completed its effectiveness study for crofelemer (Canalevia-CA1) in treating chemotherapy-induced diarrhea in dogs—a required step toward securing full FDA approval beyond its current conditional status. With topline results expected within a month and plans to submit findings in Q2 2026, this update adds a near-term development to watch alongside the company’s broader pipeline. The announcement comes as (JAGX) has already tapped $0.45 this morning, marking an approximate 18% move from yesterday’s $0.38 range, bringing early attention to the name as this news circulates. (JAGX) is now lighting up multiple bullish signals on TradingView’s technical analysis tool, as increased activity begins drawing in technical traders. If you missed my earlier coverage, keep reading to quickly get up to speed on why we’re so excited to be highlighting (JAGX) today. ===== At Krypton Street, we look for those rare inflection points—when a small biotech begins to shift from a narrow focus into something much broader. Those moments don’t happen often, but when they do, they tend to show up all at once: an established commercial base, late-stage clinical programs, fresh strategic capital, and a sharper operational direction. That’s when a story can quickly move from being overlooked to firmly on the radar. The biopharmaceutical landscape is currently witnessing that kind of evolution as Jaguar Health, Inc. (Nasdaq: JAGX) transitions from a niche provider of HIV-related supportive care into a more expansive player in gastrointestinal health and oncology supportive care. And this is exactly why (JAGX) just hit our radar and is topping today’s watchlist—Wednesday, April 8, 2026. But keep in mind, (JAGX) has less than 3M shares listed as available to the public. When companies have small public floats like this, the potential exists for big moves if demand begins to shift. In fact, First Berlin Equity Research GmbH has recently initiated coverage on (JAGX) with a $1 target, which suggests over 160% upside potential from its current $.38 range. 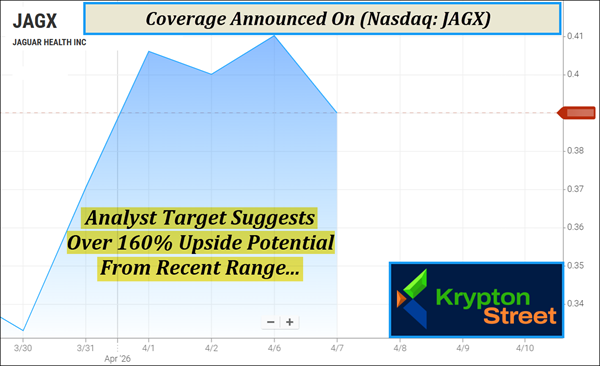
Headquartered in San Francisco, the company has built its entire therapeutic platform around crofelemer, a first-in-class anti-secretory agent that represents a paradigm shift in how chronic, debilitating diarrhea is managed. Unlike traditional antimotility agents that simply slow down the digestive system—often causing secondary complications—crofelemer works by normalizing the flow of excess chloride and water in the GI tract. The Scientific Foundation: Sustainable Botanical Medicine
At the heart of (JAGX)’s value proposition is a deep commitment to botanical medicine and environmental sustainability. Crofelemer is sustainably derived from the red bark sap of the Croton lechleri tree, found primarily in the Amazon Rainforest. The company has established a robust, patented supply chain that involves the sustainable harvesting of this sap, ensuring that the source trees remain healthy and productive for decades. This ESG-centric approach is increasingly rare in the pharmaceutical sector and provides a unique "moat" that is difficult for synthetic competitors to replicate. The product is currently FDA-approved and marketed under the brand name Mytesi®. Its primary indication is for the symptomatic relief of non-infectious diarrhea in adults with HIV/AIDS on antiretroviral therapy (ART). While this has provided a consistent revenue stream, the company’s 2026 trajectory is defined by its move into significantly larger global markets where the unmet medical need for non-opioid GI relief is substantial. The Oncology Frontier: The "OnTarget" Phase 3 Trial
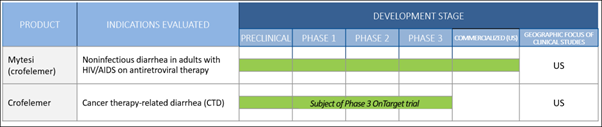
The single most significant catalyst for Jaguar Health in 2026 is the expansion of crofelemer into the oncology market. For many cancer patients, the side effects of modern targeted therapies—such as tyrosine kinase inhibitors (TKIs)—are as debilitating as the disease itself. Chemotherapy-induced overactive bowel (CIOB) and chronic diarrhea often lead to "dose holidays" or the complete discontinuation of life-saving cancer treatments. The company is currently awaiting top-line data from its Phase 3 OnTarget trial, which is evaluating crofelemer for the preventative treatment of CIOB in patients with breast or lung cancer. This is a binary event of immense proportions. Management has confirmed that the results are expected in the second quarter of 2026. If the data proves successful, (JAGX) will have the evidence needed to file for a supplemental New Drug Application (sNDA), potentially moving into an oncology supportive care market that is orders of magnitude larger than the HIV/AIDS segment. Rare Disease and Pediatric Strategy: The Intestinal Failure Pipeline
Parallel to its oncology efforts, Jaguar Health is aggressively pursuing high-value "Orphan" indications for its lead asset, crofelemer, through its subsidiaries Napo Pharmaceuticals and Napo Therapeutics.
Following a landmark $16M licensing deal with Future Pak in January 2026, the company has officially pivoted its internal R&D resources to focus exclusively on these ultra-rare intestinal failure diseases.
- Microvillus Inclusion Disease (MVID): This is a lethal, ultra-rare pediatric genetic disorder that causes life-threatening secretory diarrhea and intestinal failure. Crofelemer holds Orphan Dr-ug Designation from the FDA and EMA for MVID. Recent clinical results in pediatric patients were groundbreaking, showing a reduction in weekly parenteral support (PS) of up to 37%. Jaguar plans to file an NDA for MVID in the first half of 2027.
- Short Bowel Syndrome (SBS): The company is evaluating a highly concentrated powder-for-oral-solution formulation of crofelemer for SBS-related intestinal failure. Early data from investigator-initiated trials showed a 15.6% reduction in parenteral support needs, providing critical proof-of-concept for this billion-dollar orphan market.
- Congenital Diarrheal Disorders (CDD): Jaguar is leveraging crofelemer’s unique anti-secretory mechanism to address these heterogeneous genetic disorders. The strategy aims to reduce the "medical tap" of fluid loss, potentially qualifying the company for a high-value Priority Review Voucher (PRV) upon future regulatory approval.
Expanding the Botanical Pipeline

(JAGX) is far from a "one-product" wonder. Through its subsidiary Napo Pharmaceuticals, the company is advancing NP300, a second-generation botanical drug candidate specifically designed for the symptomatic relief of chronic diarrhea in patients with Inflammatory Bowel Disease (IBD). This expansion into the IBD space addresses a massive global market where existing treatments often prioritize reducing inflammation but fail to adequately manage the debilitating functional GI symptoms that severely diminish a patient's quality of life. Further diversifying the ecosystem, (JAGX)’s joint venture, Magdalena Biosciences, is leveraging a proprietary library of over 2,300 medicinal plants to develop novel prescription medicines for mental health indications such as ADHD and depression. Most recently, in March 2026, Magdalena initiated a preclinical study of a whole leaf coca extract for appetite suppression. Finally, the company's animal health business remains a strategic cornerstone of its broader ecosystem. Its lead veterinary drug, Canalevia-CA1, recently received an FDA conditional approval renewal through late 2026 for the treatment of chemotherapy-induced diarrhea in dogs. By applying its human GI expertise to the veterinary market, (JAGX) is able to generate incremental revenue while cross-validating its anti-secretory therapeutic approach across multiple species. Financial Position and Strategic Momentum
(JAGX) entered 2026 with several developments that underscore its evolving strategic direction. For full-year 2025, the company reported net revenue of $11.8M, with Q4 2025 net revenue rising 5% sequentially to about $3.2M. In January 2026, (JAGX) also secured a $16M upfront non-dilutive payment through its U.S. license agreement with Future Pak, with another $2M due upon completion of post-closing conditions, along with $3.0M received toward up to $20M in milestone and other future payments. This agreement shifts commercial responsibility for Mytesi and Canalevia-CA1 to Future Pak, allowing Jaguar to sharpen its focus on late-stage crofelemer development in human rare-disease intestinal failure indications. Jaguar has also noted that Future Pak has already purchased product from the company, an early sign that the new commercial structure is beginning to take shape. Together, these developments highlight a company that is strengthening its financial footing while aligning its resources around what management sees as its most promising path forward. Strategic Corporate Outlook
As we approach the mid-point of 2026, (JAGX) sits at the intersection of proven commercial success and explosive clinical potential. The transition to a "covered" status by major payers for Mytesi® has provided a case study in how to navigate the complex U.S. reimbursement landscape for botanical drugs. Management is now looking to apply that same playbook to the oncology and rare disease sectors. The upcoming months are expected to be the most active in the company's history. 7 Reasons Why (JAGX) is Topping Our Watchlist This Morning —Wednesday, April 8, 2026…1. Analyst Coverage: First Berlin Equity Research GmbH recently initiated coverage on (JAGX) with a $1 target, which suggests over 160% upside potential from this week’s $0.38 range.
2. Small Float: With fewer than 3M shares listed as available to the public, (JAGX)’s small float could witness the potential for big movers if demand begins to shift. 3. Phase 3 Catalyst: (JAGX) is awaiting top-line data in Q2 2026 from its OnTarget Phase 3 study in cancer supportive care, giving the company a major near-term event to watch. 4. Approved Product: (JAGX) already has an FDA-approved product, Mytesi, which means the company is working from an existing commercial base rather than starting from zero. 5. Fresh Capital: In January 2026, (JAGX) secured a $16M upfront non-dilutive payment from Future Pak, with another $2M due after post-closing conditions and $3.0M received toward up to $20M in milestone and other future payments. 6. Revenue Trend: (JAGX) reported $11.8M in full-year 2025 net revenue, and Q4 2025 net revenue rose 5% sequentially to about $3.2M, showing measurable commercial traction. 7. Rare Disease: (JAGX) is focusing its internal R&D efforts on ultra-rare intestinal failure conditions, including MVID, where recent pediatric results showed up to a 37% reduction in weekly parenteral support. Put (JAGX) On Your Screen While It’s Still Early…

What makes (JAGX) stand out right now is how multiple pieces are coming together at once. A small float, recent analyst coverage, and a clearly defined near-term Phase 3 readout in Q2 2026 create a setup that continues to draw attention. At the same time, the company is not starting from scratch—Mytesi is already on the market, revenue reached $11.8M in 2025 with sequential growth into Q4, and a $16M upfront agreement with Future Pak adds meaningful non-dilutive capital while sharpening focus on rare intestinal failure programs like MVID, where early data has shown measurable reductions in parenteral support. Put it all together, and (JAGX) is a name where structure, clinical timing, and strategic execution are all aligning in a way that makes it worth a closer look. We’re watching (JAGX) closely this morning. Give (JAGX) a look before the day gets moving. And stay tuned for my next update, it could be on its way to you very shortly. Sincerely, Alex Ramsay
Co-Founder / Managing Editor Krypton Street Newsletter
|





No comments:
Post a Comment