Any content you receive is for information purposes only. Always conduct your own research. *Disseminated on Behalf of Medicus Pharma Ltd.
Krypton Street Just Placed (NASDAQ: MDCX) At The Top of Tomorrow’s Watchlist—Wednesday, May 6, 2026 Don’t Miss The Next Breakout—Get Real-Time Alerts Sent Directly To Your Phone. Up To 10X Faster Than Email.
We’ll Begin Covering (MDCX) Early
Take A Look At (MDCX) Before Tomorrow Morning…
May 5, 2026
We'll Have All Eyes on (Nasdaq: MDCX) Tomorrow Morning — Here's Why
Dear Reader, Krypton Street is bringing a familiar name back into focus: Medicus Pharma Ltd. (NASDAQ: MDCX). We’ve covered (MDCX) before—but recent developments are putting it back on our radar in a big way. The company continues advancing SkinJect®, its localized microneedle platform designed to target basal cell carcinoma lesions directly, offering a potential alternative for patients dealing with recurring or multiple tumors. Updated Phase 2 data has reinforced clinical clearance at higher dose levels, while a new FDA-facing orphan designation submission for Gorlin Syndrome introduces a rare-disease pathway that wasn’t part of the story before. Add in recent Capitol Hill engagement around accelerating access, and the setup has shifted enough for Krypton Street to take another close look. That’s why (MDCX) is back at the top of our watchlist heading into tomorrow morning—Wednesday, May 6, 2026 But keep in mind, one analyst has a target on (MDCX) that you’ll want to see. Jason McCarthy, Ph.D., Senior Managing Director, Head of Biotechnology Research at Maxim Group, has a $2 target on (MDCX) which suggests 700% upside potential from its recent $.25 range. 
Headquartered in Philadelphia, Medicus Pharma Ltd. (NASDAQ: MDCX) operates with a distinct capital-efficient model designed to accelerate the clinical programs of disruptive therapeutic assets. Rather than bearing the massive costs of late-stage global commercialization, the company’s stated strategy is to advance select candidates through Phase 2 clinical proof-of-concept and subsequently pursue high-value licensing or strategic partnerships with established pharmaceutical firms. This model is currently being tested through two primary therapeutic pillars: the SkinJect™ platform for dermatology and oncology, and the Teverelix hormone-suppression program. Dermatology Pillar: SkinJect™ and the D-MNA Platform
The company's lead asset, SkinJect™ (D-MNA), is a novel localized immuno-oncology precision product. It utilizes a patented dissolvable microneedle patch to deliver doxorubicin directly into basal cell carcinoma (BCC) lesions. This non-invasive delivery method aims to provide a viable alternative to surgical resection, which can often be disfiguring for patients with multiple or recurrent lesions. 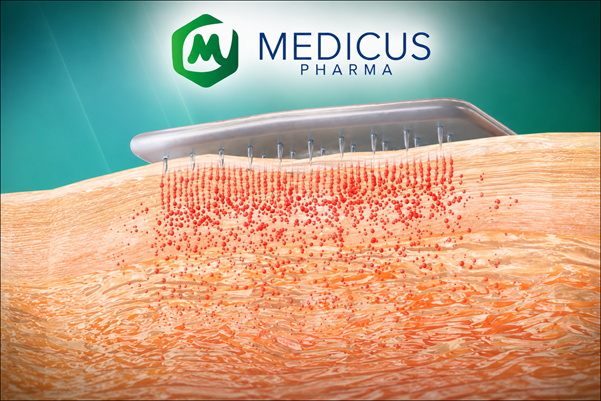
- Phase 2 Milestones: In December 2025, (MDCX) successfully completed enrollment of 90 patients in its U.S.-based randomized, double-blind Phase 2 study (SKNJCT-003).
- Topline Data Validation: Topline results released in early 2026 revealed highly promising results, particularly in the highest dose (200ug) cohort, which showed up to a 73% clinical clearance rate.
- Surgical Avoidance: Key opinion leaders have highlighted that the data suggests approximately 3 out of 4 patients treated with the optimal dose may avoid immediate surgery.
- Regulatory Path: Following a Type C meeting with the FDA, the company is pursuing a potential 505(b)(2) regulatory pathway, which could significantly compress the time to market.
Hormone-Suppression Pillar: Teverelix
In August 2025, (MDCX) significantly expanded its portfolio by acquiring Antev, a UK-based biotech developing Teverelix. Teverelix is a next-generation long-acting GnRH antagonist designed to suppress sex hormone production without the initial "testosterone flare" associated with legacy GnRH agonists.
The Agentic AI Strategy: Scaling Through Simulation

A unique differentiator for (MDCX) is its integration of agentic AI-driven clinical development capabilities. (MDCX) entered into a collaboration with Reliant AI Inc. to develop a platform optimized for:
- Protocol Simulation: Testing trial designs in a virtual environment before patient enrollment.
- Dynamic Site Selection: Identifying geographic locations with the highest concentration of target patient populations to accelerate enrollment.
- Patient Stratification: Utilizing pharmacodynamic-informed data to ensure the most responsive participants are prioritized, thereby de-risking the clinical outcome.
Financial and Market Metrics
(MDCX) enters the second quarter of 2026 with a market capitalization of approximately $16M and a cash position of $8.7M as of Q2 2026. During fiscal year 2025, the company successfully raised approximately $31.9M in total financing proceeds to fund its accelerated Phase 2 programs. While the company currently reports a net loss consistent with high-growth biotech development ($47.3M for FY2025), management emphasizes that their capital-efficient model is focused on near-term value creation through proof-of-concept readouts rather than a long-term cash-intensive commercial build-out. Latest Headlines
April 27, 2026
Medicus Pharma CEO meets with lawmakers on Energy & Commerce Committee on Capitol Hill
April 17, 2026
Medicus Pharma Submits Orphan Designation Application to U.S. FDA for SkinJect® in Gorlin Syndrome
April 06, 2026
Medicus Pharma Submits Optimized Phase 2 Study Protocol to U.S. FDA for Teverelix in Acute Urinary Retention
April 01, 2026
Medicus Pharma Ltd Clarifies Positive SkinJect Phase 2 Dataset Expanded April 2026 Program UpdatesSkinJect® Orphan Designation Expansion
(MDCX) submitted an Orphan Designation application to the U.S. FDA for SkinJect® in basal cell carcinoma patients with Gorlin Syndrome, a rare genetic disorder associated with multiple, recurring skin cancers. This represents a strategic expansion of SkinJect into a high-unmet-need orphan indication where no FDA-approved therapies currently exist and patients often rely on repeated surgical procedures. Because SkinJect is designed as a localized, repeatable, non-surgical microneedle-based treatment, the program may be especially relevant for Gorlin Syndrome patients who can develop dozens to hundreds of lesions over time. If granted, orphan designation could provide regulatory and commercial advantages, including potential market exclusivity, tax credits, fee waivers, and enhanced FDA interaction. Capitol Hill Engagement & Regulatory Acceleration Strategy
(MDCX) also announced that CEO Dr. Raza Bokhari and the company’s leadership team met with lawmakers on Capitol Hill, including members of the House Energy & Commerce Committee, to build support for accelerated access to SkinJect® for Gorlin Syndrome patients. The discussions focused on orphan designation, a potential registrational IND pathway, and rare disease pediatric voucher opportunities. In collaboration with the Gorlin Syndrome Alliance, (MDCX) emphasized the lifelong burden faced by patients who often experience recurring basal cell carcinomas and have limited non-surgical treatment options. This policy engagement adds another layer to the SkinJect strategy by combining clinical development, regulatory planning, patient advocacy, and legislative outreach around a rare disease pathway. 7 Reasons Why Krypton Street Will Have All Eyes On (MDCX) Tomorrow Morning—Wednesday, May 6, 2026…
1. Under The Radar: with an approximate $16M market capitalization and recent $.25 range, (MDCX) appears to be a little-known biotech name with multiple recent clinical and regulatory updates worth keeping an eye on. 2. Analyst Target: one Maxim Group analyst has a $2 target on (MDCX), suggesting 700% upside potential from its recent $.25 range. 3. SkinJect Data: recent Phase 2 results for (MDCX) showed up to 73% clinical clearance and 40% histological complete response at Day 57 in the 200µg cohort. 4. Orphan Filing: the latest FDA-facing orphan designation application gives (MDCX) a new rare-disease angle through SkinJect® in Gorlin Syndrome. 5. Capitol Hill: recent meetings with lawmakers add a policy and access-focused layer to the (MDCX) SkinJect® strategy. 6. Teverelix Clearance: FDA “study may proceed” clearance allows (MDCX) to advance a Phase 2b Teverelix study in advanced prostate cancer patients with high cardiovascular risk. 7. AI Collaboration: the Reliant AI collaboration gives (MDCX) a clinical-development angle tied to protocol simulation, site selection, and patient stratification. Take A Look At (MDCX) Before Tomorrow Morning…
When you step back and connect the pieces, a broader picture begins to take shape. A clinical program delivering up to 73% clearance at higher doses… a second program advancing with FDA clearance into Phase 2b… and a rare-disease pathway now in motion through an orphan designation submission. Layer in policy engagement at the federal level, a collaboration focused on optimizing clinical development through AI, and a capital structure that places the company in a little-followed category, and you begin to see multiple drivers unfolding at once. Add to that a published analyst target suggests 700% upside potential from recent levels, and it becomes clear why this name is starting to surface on more screens. From clinical progress to regulatory positioning and strategic execution, (MDCX) is building a story with several moving parts—each worth understanding as new developments continue to emerge. (MDCX) will be front and center for us tomorrow morning. Take a few minutes to review it tonight. Sincerely, Alex Ramsay
Co-Founder / Managing Editor Krypton Street Newsletter
|




No comments:
Post a Comment